| References: |
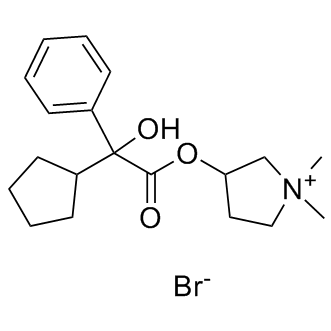
Glycopyrrolate(Glycopyrronium Br) is a muscarinic competitive antagonist used as an antispasmodic.in vitro: Glycopyrrolate showed no selectivity in its binding to the M1-M3 receptors. Kinetics studies, however, showed that glycopyrrolate dissociates slowly from HASM muscarinic receptors (60% protection against [3H]-NMS binding at 30 nM) compared to ipratropium bromide.
in vivo: Glycopyrrolate (1 mg) tablets were then administered, starting with one tablet daily the third week and increasing the daily dose by one tablet per week until a maximum of four tablets during week six and 4 days of week seven when the daily dose was reduced to two tablets for 3 days. glycopyrrolate can be given in controlled doses provided that an adequate medical assessment has been undertaken. Glycopyrrolate has a slow and erratic absorption from the gastrointestinal system, but even low plasma levels are associated with a distinct and long-lasting antisialogic effect. Oral glycopyrrolate is emerging as a potential second-line treatment option, but experience with safety, efficacy, and dosing is especially limited in children. phase III study, 52.3% of glycopyrrolate oral solution recipients (aged 3-18 years; n = 137) had an mTDS response (primary endpoint); the response rate was consistently above 50% at all 4-weekly timepoints, aside from the first assessment at week 4 (40.3%). In general, glycopyrrolate oral solution was well tolerated in clinical trials. The majority of adverse events were within expectations as characteristic anticholinergic outcomes. |